When consuming iced beverages, it casually seemed that ice would disappear at different rates depending on the beverage. What happens when we actually test this?
Initial observation/Question:
It seemed that when drinking iced beverages, ice would melt faster in a glass of water than in brandy. This led us to question if ice actually melts faster in water or brandy.
Expectation:
Uncontrolled observation suggested that ice would disappear quite quickly in a glass of water compared to a glass of brandy, so that is the starting expectation.
Equipment & Materials:
- 2 similar glasses
- 2 measuring glasses
- 3 oz brandy (80 proof)
- 3 oz water
- ice cubes (6)
- scale (we used a AMW-2000 Kitchen Scale
)
- probe thermometer (ThermoWorks Thermapen
)
- camera with time-lapse capability (optional, e.g. iPhone with iOS 8)
Procedure:
- weigh and tare a measuring glass
- measure and weigh 3 oz of water (read to the bottom of the meniscus), remove from scale and set aside
- weigh and tare the glass
- add 3 ice cubes
- record mass of ice
- repeat steps 1-5 with brandy and the 2nd glass
- measure starting temperature of the brandy and water
- start time-lapse camera if using
- add fluids to ice at the same time
- measure temperature of fluids occasionally as ice melts
Data:
3 oz brandy = 88.3 g; ice (brandy) = 65.1 g
3 oz water = 91.3 g; ice (water) = 67.7 g
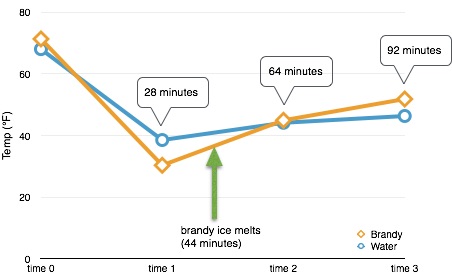
| Brandy Temp (°F) | Water Temp (°F) | Notes | |
| time 0 | 71.3 | 68.0 | |
| time 1 | 30.3 | 38.5 | |
| time 2 | 44.9 | 44.1 | all ice in brandy is melted |
| time 3 | 51.8 | 46.3 | water has minuscule ice |
The timelapse video is 30 frames per second, 23 seconds long, and represents 1 frame acquired every 8 seconds. The video runs between actual (clock) times of ~9:20 – 11pm. For a description of how timelapse on iOS 8 works, please see here.
Surprise Results:
Leaving brandy on the counter is a fairly good way to test if your kitchen is home to any fruit flies…
Conclusion:
Despite what casual observation indicated, ice melts significantly faster in brandy than in water. From a chemical standpoint this makes sense. What is brandy but water with stuff (solutes) in it? Solutes in water disrupt ice formation and encourage melting. See here for an interactive demo. We happen to use salt on the roads and walkways in areas that have snowy winter climates because it is abundant and cheap, but sugar and alcohol work the same way… it would just be very expensive, and possibly a bit sticky later, to hose everything down with brandy.
While the water did start at a colder temperature and have slightly more ice due to variations in ice cube mass, the brandy reached a lower temperature as part of the cooling curve, so it is considered doubtful that these initial variations impacted the outcome. It is suspected that the initial observation was based on the fact that we never pour ourselves 3 oz of water. Typically, a glass of water in our household is somewhere between 15-28 oz.
Future Questions:
How would varying the proof of alcohol impact ice melting rates? Would working with a typical serving of water vs a typical pour of brandy match the previously observed behavior (in which case, how much more water is needed than brandy to have equal melting rates)?
Related Works:
Provost, Dan. Sept 23, 2014. “How Does the iOS 8 Time-lapse Feature Work?”. Studio Neat. <http://www.studioneat.com/blogs/main/15467765-how-does-the-ios-8-time-lapse-feature-work> accessed Nov 16, 2014.
Senese, Fred. Feb 10, 2010. “Why does salt melt ice?”. General Chemistry Online! <http://antoine.frostburg.edu/chem/senese/101/solutions/faq/why-salt-melts-ice.shtml> accessed Nov 16, 2014